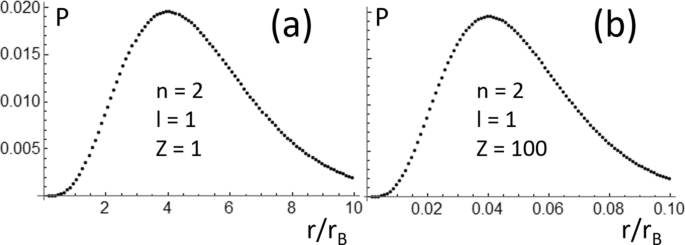
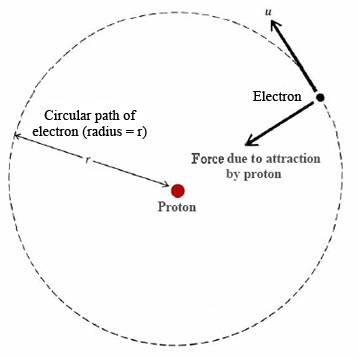
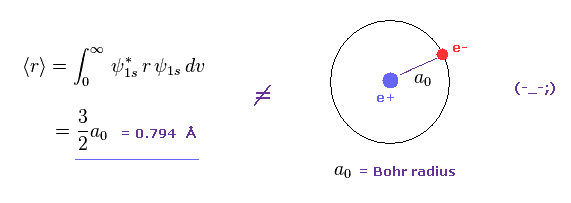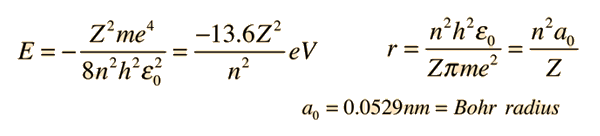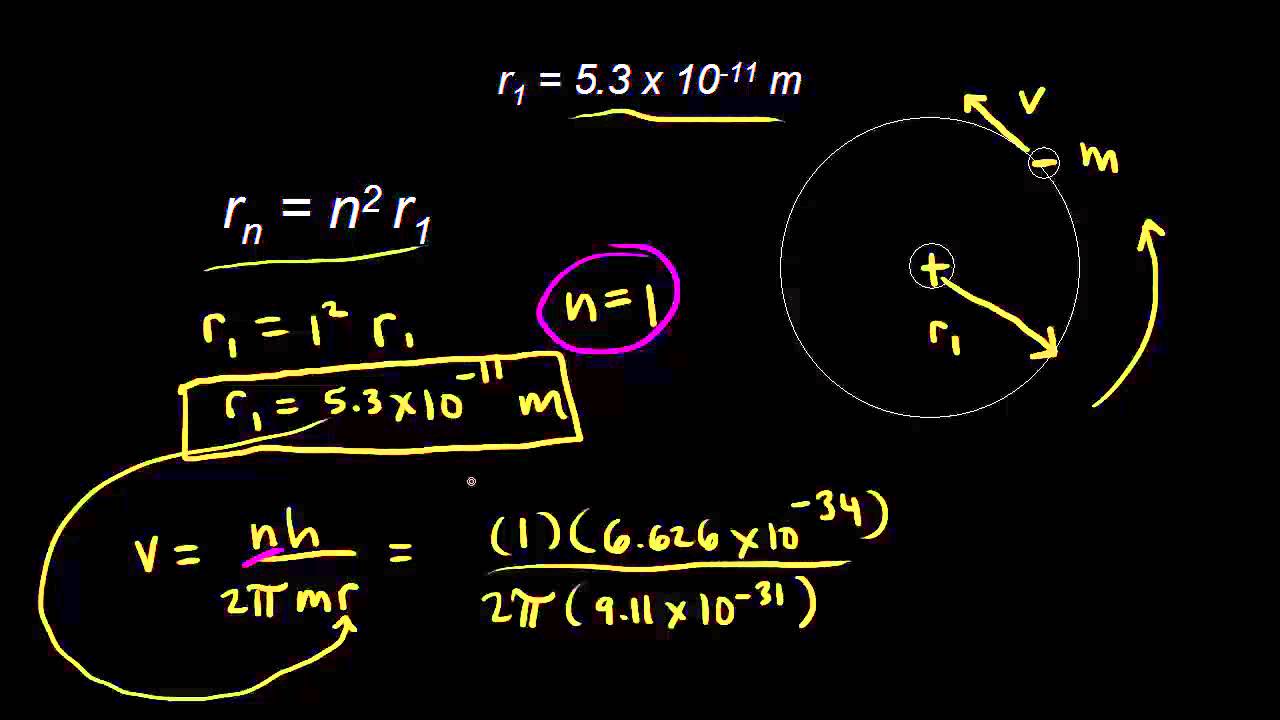10. The value of Bohr's radius of hydrogen atom is : 0.529 x 10 cm 60.529 x 10-10 cm c) 0.529 x 10-12 cm d) 0.529 x 10-6 cm
Calculate radius of second Bohr radius of hydrogen atom and then also calculate the speed of the electron and total - Sarthaks eConnect | Largest Online Education Community

MathType on Twitter: "The most probable distance between the nucleus and the electron in a hydrogen atom in its ground state is given by the Bohr Radius. This physical constant is named

Bohr radius of small hydrogen atom as a function of orbital quantum... | Download Scientific Diagram
Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community

The radius of the second Bohr orbit for hydrogen atom is: [Given: Planck's constant. h=6.6262×10^−34 Js; mass of electron = 9.1091×10^−31kg; charge of electron, e=1.60210×10^−19 C; permittivity of vacuum, (ε 0) =

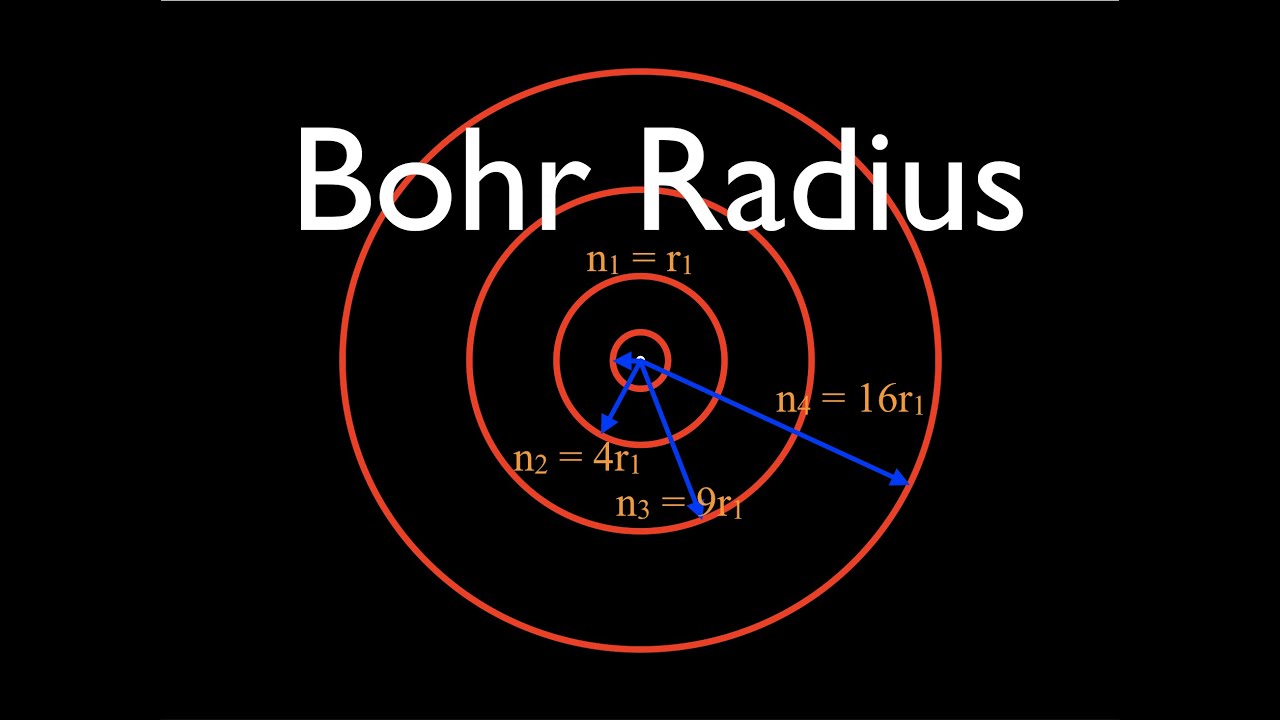
Using Bohr's postulates of atomic model, derive the expression for radius of nth electron orbit. Hence, obtain the expression for Bohr's radius. | Snapsolve

1. The first Bohr's radius (for electron in hydrogen atom in the ground state): 2. The ground energy level in hydrogen atom: - ppt video online download
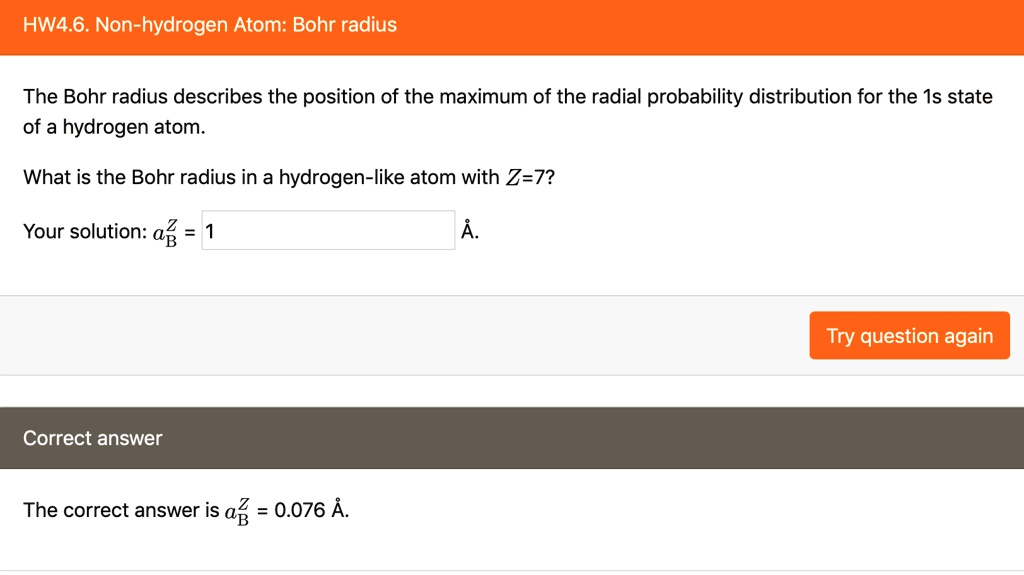


.PNG)